Designed For You
Your New Go-to Bladder Scanner
- Eliminate volume measurement uncertainty with precision detection and intuitive guidance
- Ensure accurate readings every time – even in challenging cases where traditional bladder scanners fail.
- Say goodbye to over- and underestimation issues that plague other devices
- Trust in reliable results that enhance patient care
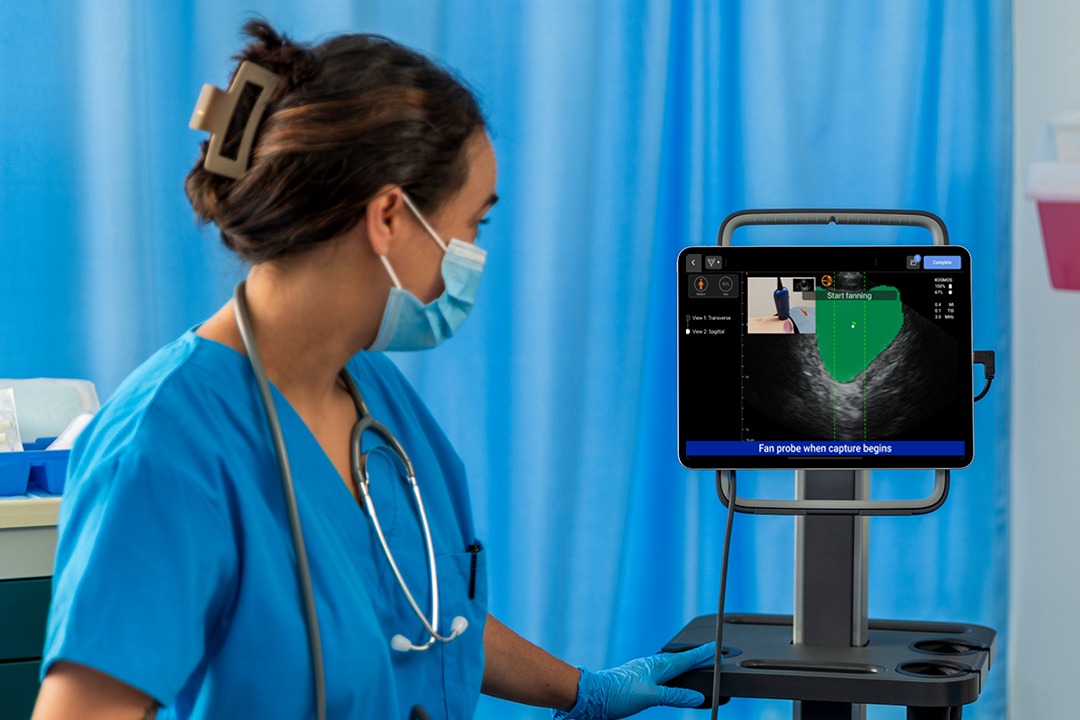
Reliability You Can Count On
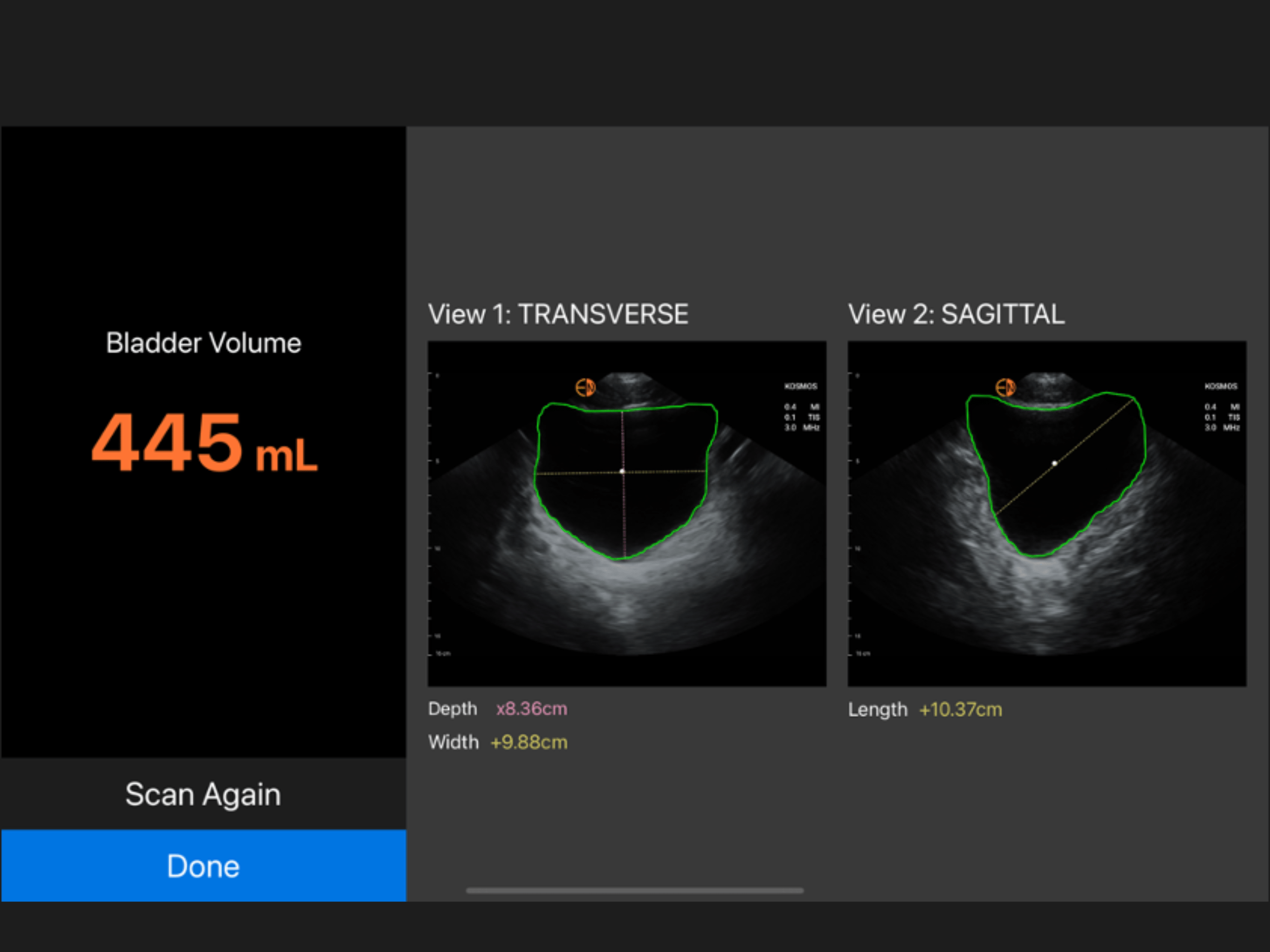
Kosmos Bladder represents a groundbreaking ultrasound solution that sets a new standard for bladder volume measurement.

Kosmos Bladder AI Workflow
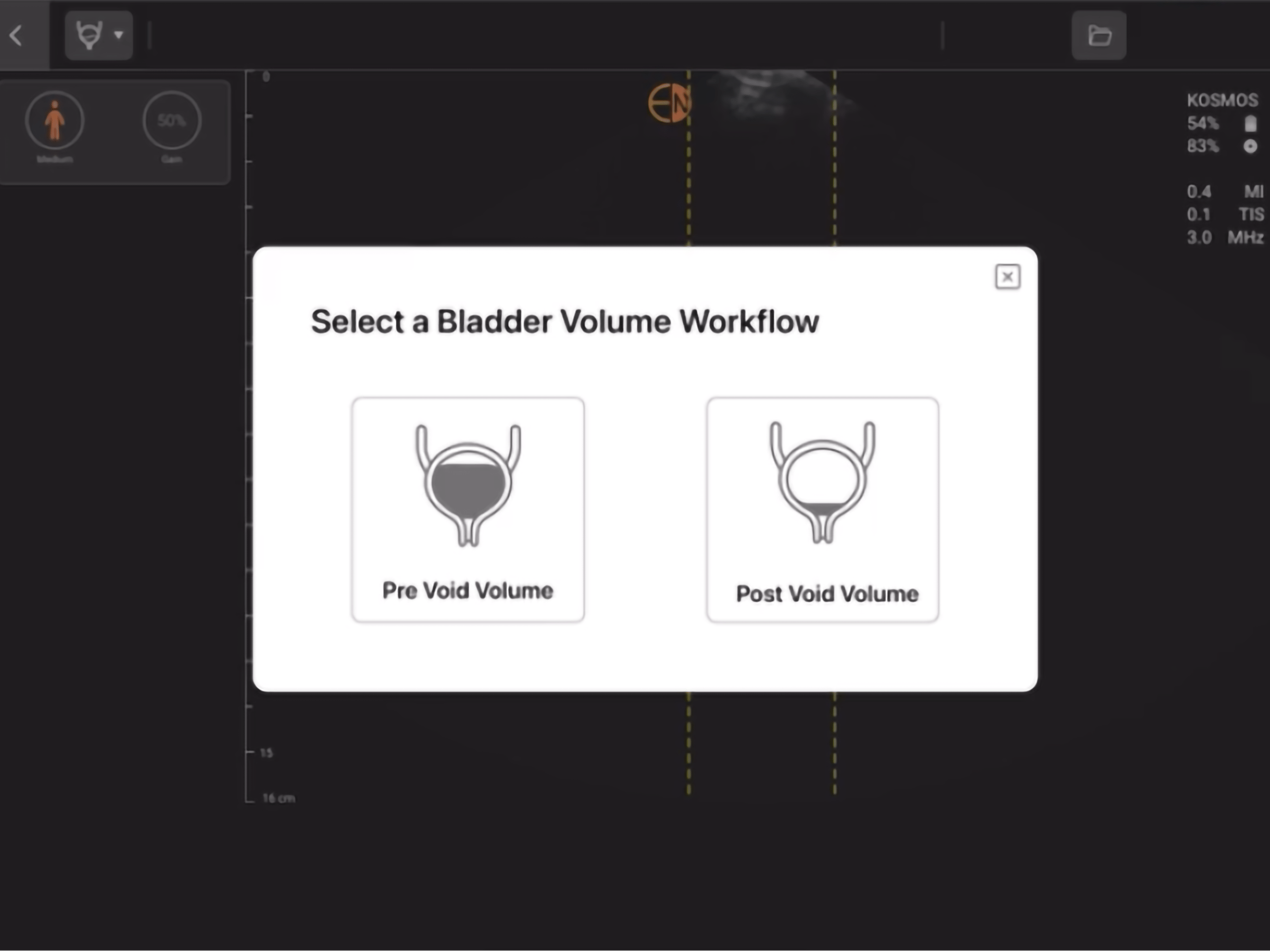
Dynamic AI that detects pre- and post void bladder volume.

Clinically Validated Accuracy
With clinically validated accuracy for increased confidence.

Quality You Can Trust
Robust 5-year warranty, with military-grade transducers that withstand 1m drops and extreme environmental conditions.
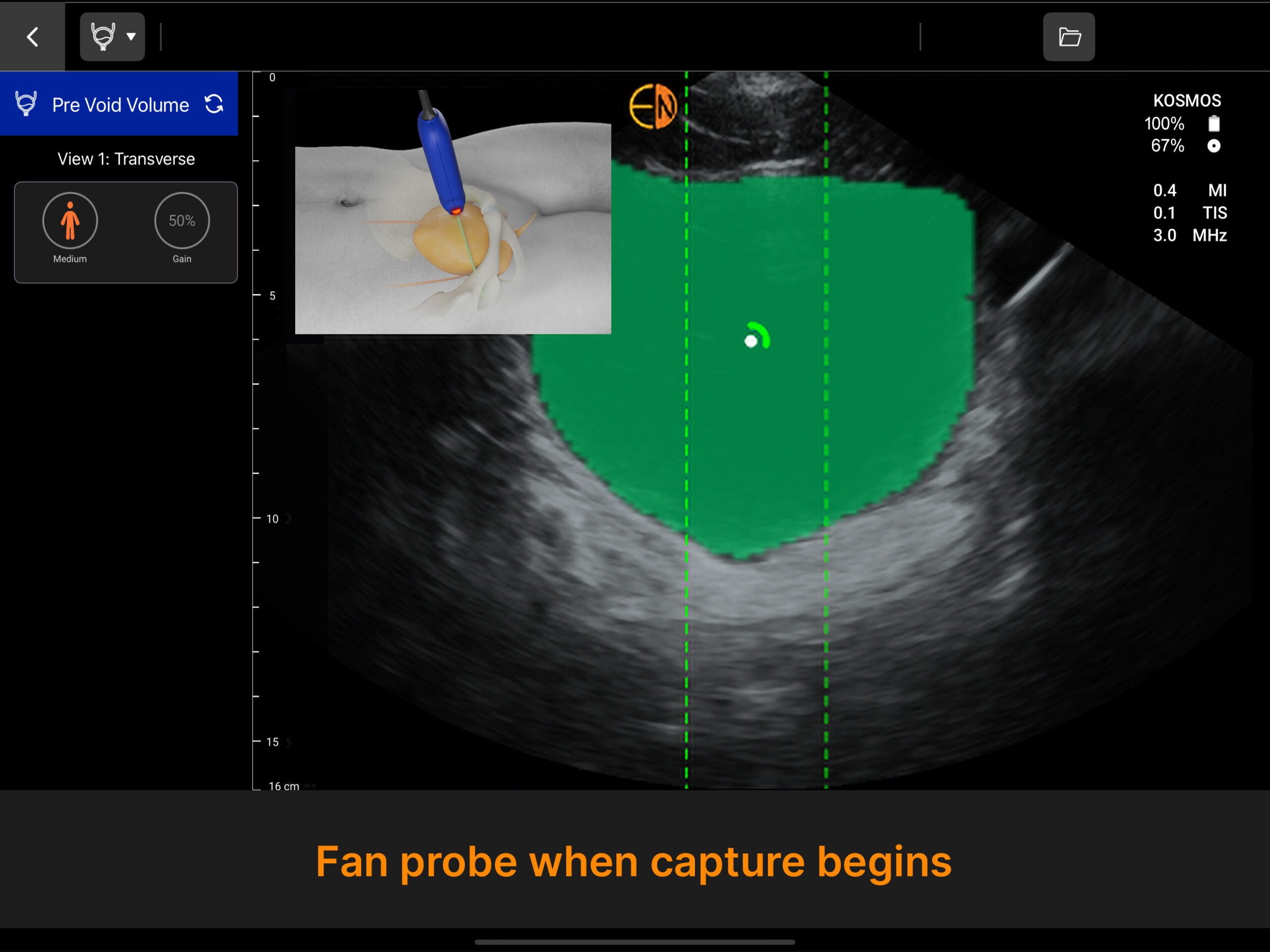
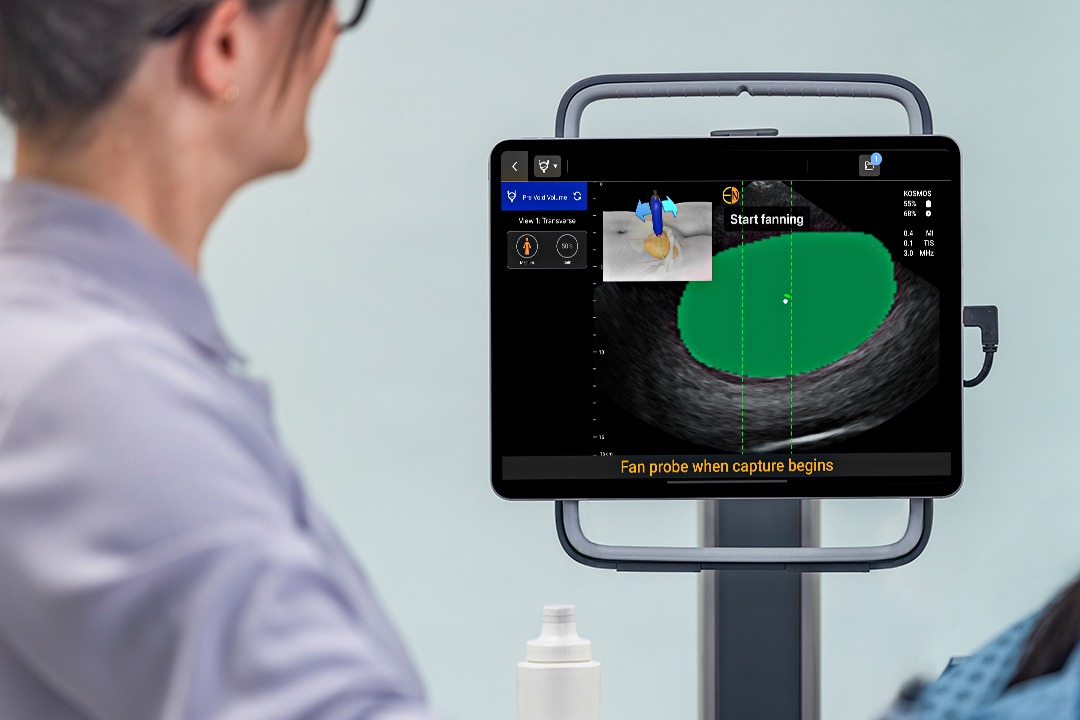
Powered by the advanced AI technology of Kosmos, the Kosmos Bladder scanner sets a new standard for accuracy, reliability, and user-friendly design—all while remaining affordable for any budget. Use it as a dedicated bladder scanner or expand its capabilities for an all-in-one POCUS solution.
Request Information
More Than Just A Bladder Scanner
Expand Kosmos Bladder into a comprehensive Point-of-Care Ultrasound solution, with an intuitive interface and AI-driven technology, capable of replacing three separate systems. Offering bladder, vascular access, and multiple POCUS applications, Kosmos Bladder delivers an efficient, versatile medical imaging tool designed to empower healthcare professionals with accurate, reliable diagnostic capabilities.

Kosmos Torso-One
Heart | Lungs | Abdomen | Bladder
Experience comprehensive, whole-torso imaging with our sleek, small footprint Torso-one probe, featuring advanced Doppler capabilities, AI integration, and automated presets—all backed by a 5-year warranty.

Kosmos Lexsa
MSK | Nerve | Vascular | Lung
Acquire exceptional MSK, vascular, nerve, and lung imaging with the Lexsa probe’s powerful 38mm linear array technology and precision channel processing, featuring advanced, real-time Doppler capabilities and backed by a 5-year warranty.
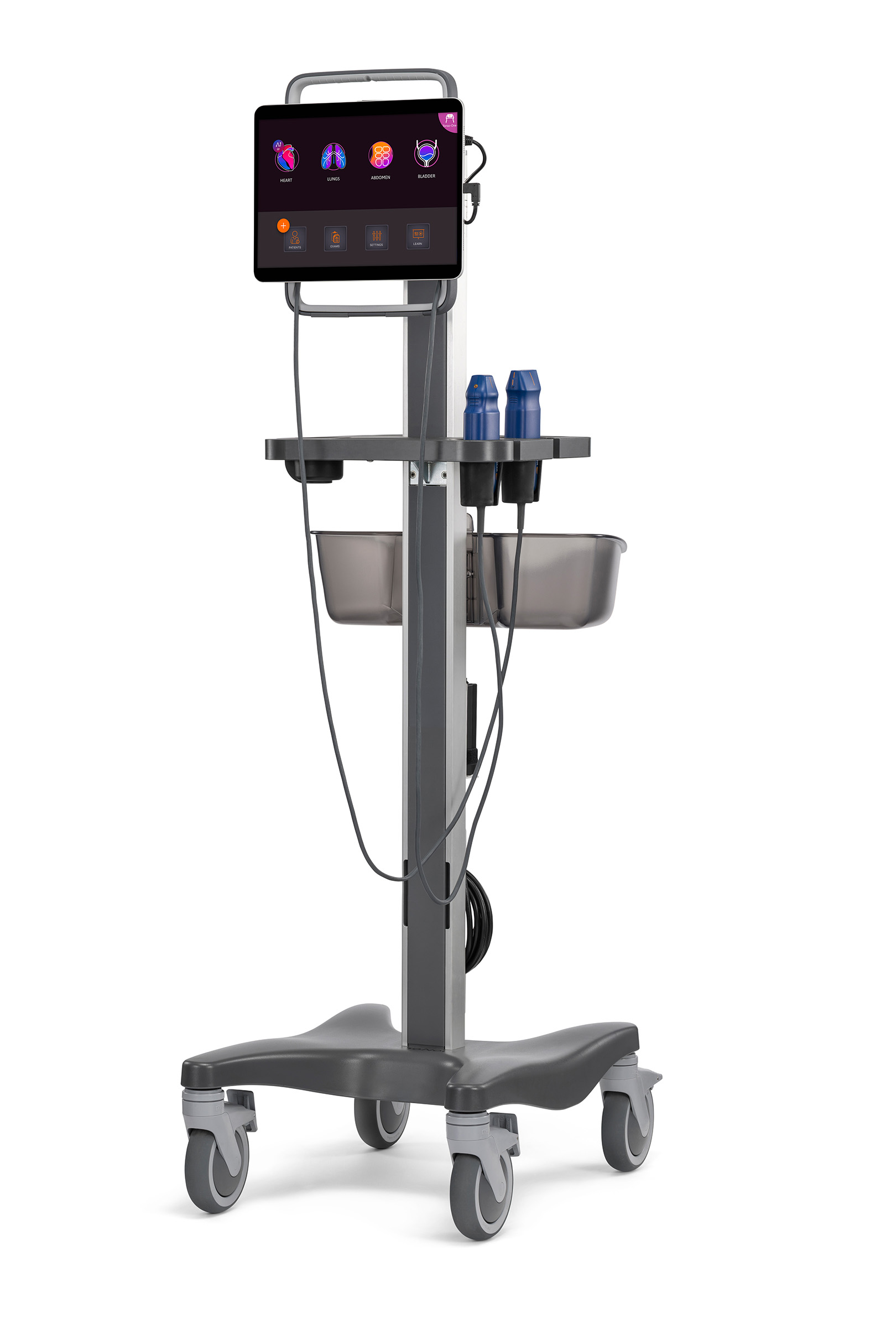
On the Stand
Tablet security
Maximize the efficiency of your patient care while keeping your equipment secure and all in one place.
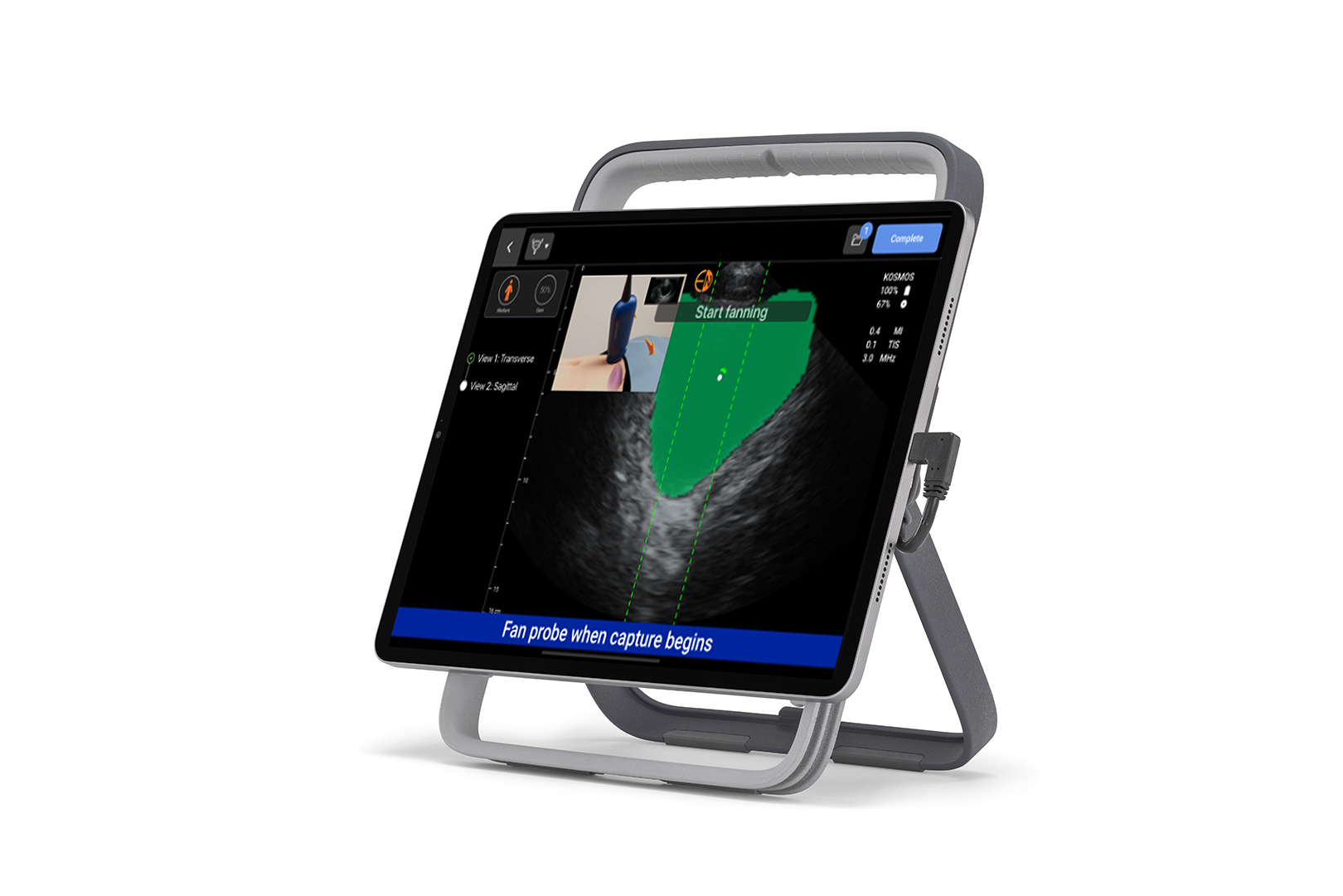
Portable
Fully detachable monitor for bedside flexibility
Accessible, and convenient configuration options, designed to fit with your existing workflow.
Reduce complications, empower accurate care decisions, and improve confidence